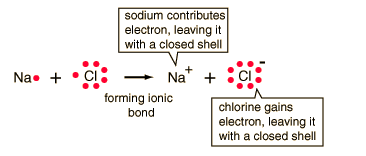
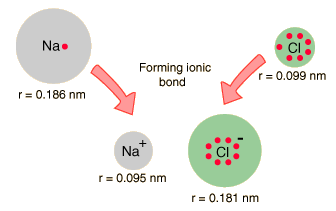
Calcium Chloride
|
Calcium chloride is a salt that is marketed in pellet form for removing dampness from rooms. Remarkably hydroscopic, it is placed in perforated containers or on screens with a containers below them. Calcium chloride will collect enough water from the air to completely transform the pellets, leaving an empty basket and a volume of solution below it which is several times the volume of the original pellets.
|
Calcium chloride is also widely used as a deicer since it is effective in lowering the melting point when applied to ice. The melting point lowering occurs with any solute and is a colligative property, being proportional to the number of solute particles. Calcium chloride is a more effective deicer than sodium chloride since each molecule produces three ions in solution compared to two for NaCl. Calcium chloride is effective in melting ice down to -25°F, wheras sodium chloride loses its effectiveness at about 20°F and is not useful for this purpose below 15°F.
Calcium chloride is used for maintaining unpaved roads and for fortifying roadbases for new construction. It is sometimes used in concrete to allow it to be worked with less water, shortening drying time. It increases compressive strength and water resistance of concrete, but diminishes flexural and tensile strength. For that reason it is not permitted in pre-stressed concrete.
|
Index
Chemistry concepts |